All Photos(1)
About This Item
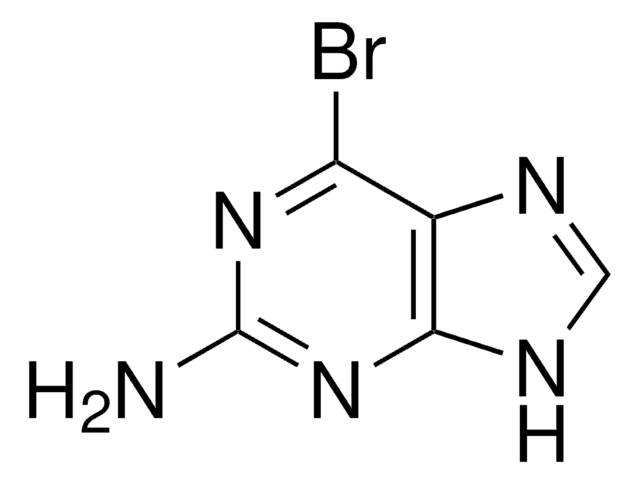
Empirical Formula (Hill Notation):
C5H3BrN4
CAS Number:
Molecular Weight:
199.01
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
assay
98%
form
solid
mp
>300 °C (lit.)
SMILES string
Brc1ncnc2nc[nH]c12
InChI
1S/C5H3BrN4/c6-4-3-5(9-1-7-3)10-2-8-4/h1-2H,(H,7,8,9,10)
InChI key
CTGFGRDVWBZYNB-UHFFFAOYSA-N
Related Categories
General description
6-Bromopurine enhances the carcinostatic activity of azaserine in a test system employing ascites cell forms of sarcoma 180 and Ehrlich carcinoma in vivo. 6-bromopurine nucleosides are excellent substrates for substitution reactions with N-, O-, and S-containing nucleophiles in polar solvents.
Application
6-Bromopurine was used in the synthesis of 6-halopurine alkynes and corresponding triazole derivatives. 6-Bromopurine was used in the synthesis and chemical characterization of 2,3,4,5-tetrahydro-1,5-benzoxazepines-3-ol.
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Comparison of some biologgical and biochemical properties of 6-bromopurine and 6-iodopurine.
A C SARTORELLI et al.
Biochemical pharmacology, 11, 1017-1024 (1962-11-01)
E A Véliz et al.
The Journal of organic chemistry, 66(25), 8592-8598 (2001-12-12)
Surprisingly facile direct substitution reactions with acetyl-protected 6-bromopurine nucleosides are described. Included in the series of bromonucleosides studied is the guanosine derivative N(2)-2',3',5'-tetraacetyl-6-bromopurine ribonucleoside, the synthesis of which is reported here for the first time. Brominated nucleosides had not previously
Synthesis, unambiguous chemical characterization, and reactivity of 2, 3, 4, 5-tetrahydro-1, 5-benzoxazepines-3-ol.
Garcia-Rubino ME, et al.
Royal Society of Chemistry Advances, 2(33), 12631-12635 (2012)
Eva Galante et al.
Molecules (Basel, Switzerland), 18(5), 5335-5347 (2013-05-15)
2-[¹⁸F]Fluoroethyl azide ([¹⁸F]FEA) can readily be obtained by nucleophilic substitution of 2-azidoethyl-4-toluenesulfonate with [¹⁸F]fluoride (half-life 110 min), and has become widely used as a reagent for 'click' labeling of PET tracers. However, distillation of [18F]FEA is typically required, which is
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service