All Photos(1)
About This Item
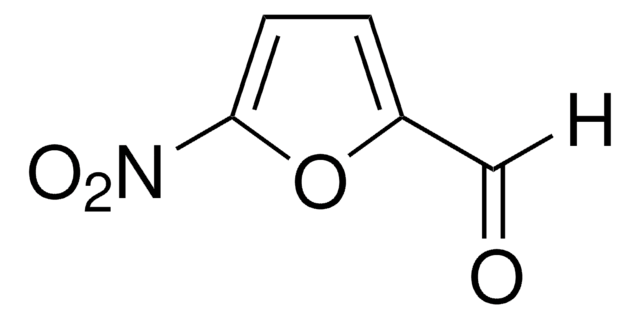
Linear Formula:
O2NC6H4CH=CHCHO
CAS Number:
Molecular Weight:
177.16
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
assay
98%
form
solid
mp
140-143 °C (lit.)
storage temp.
2-8°C
SMILES string
[H]C(=O)\C=C\c1ccc(cc1)[N+]([O-])=O
InChI
1S/C9H7NO3/c11-7-1-2-8-3-5-9(6-4-8)10(12)13/h1-7H/b2-1+
InChI key
ALGQVMMYDWQDEC-OWOJBTEDSA-N
Related Categories
General description
Reduction of trans-4-nitrocinnamaldehyde using the polymer-supported Hantzsch 1,4-dihydropyridine ester and a catalytic amount of HCl has been investigated.
Application
4-Nitrocinnamaldehyde has been used in the preparation of 2, 2′-[(E)-3-(4-nitrophenyl) prop-2-ene-1,1-diyl] bis(3-hydroxy-5, 5-dimethylcyclohex-2-en-1-one).
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Polymer-Supported Hantzsch 1, 4-Dihydropyridine Ester: An Efficient Biomimetic Hydrogen Source.
He R, et al.
Advanced Synthesis & Catalysis, 350(1), 54-60 (2008)
Joo Hwan Cha et al.
Acta crystallographica. Section E, Structure reports online, 68(Pt 8), o2510-o2510 (2012-08-21)
In the title compound, C(25)H(29)NO(6), each of the cyclo-hexenone rings adopts a half-chair conformation. The hy-droxy and carbonyl O atoms face each other and are oriented to allow for the formation of two intra-molecular O-H⋯O hydrogen bonds. In the crystal
E Eder et al.
Mutagenesis, 6(4), 261-269 (1991-07-01)
Seventeen cinnamaldehydes, cinnamic acids, 2-furylacroleins and related compounds were tested in the Salmonella preincubation reversion assay and in the SOS chromotest. Of eight compounds containing nitrogroups, seven were clearly mutagenic in the presence of S9 mix and six in its
I Baburina et al.
Biochemistry, 37(5), 1245-1255 (1998-03-07)
The residue C221 on pyruvate decarboxylase (EC. 4.1.1.1) from Saccharomyces cerevisiae has been shown to be the site where the substrate activation cascade is triggered [Baburina et al. (1994) Biochemistry 33, 5630-5635] and is located on the beta domain [Arjunan
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service