291390
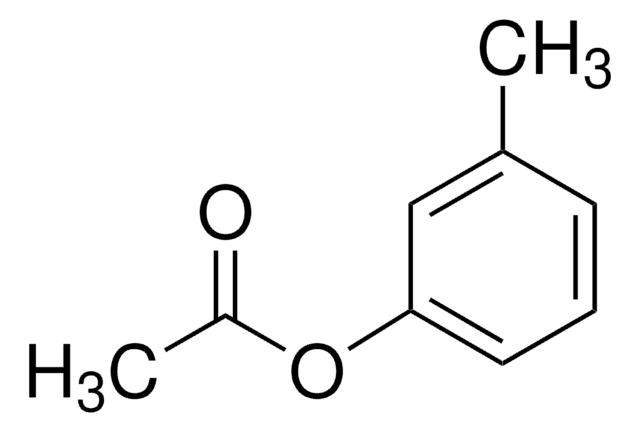
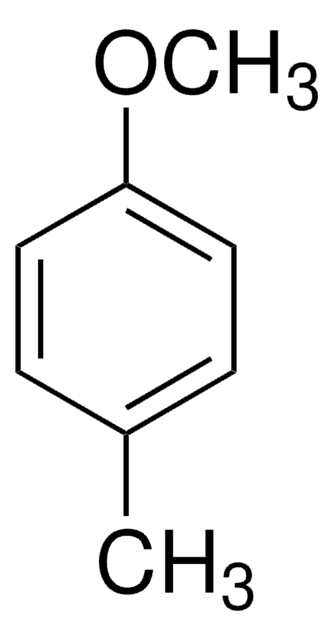
p-Tolyl acetate
99%
Synonym(s):
p-Cresyl acetate
About This Item
Recommended Products
assay
99%
refractive index
n20/D 1.501 (lit.)
bp
210-211 °C (lit.)
density
1.047 g/mL at 25 °C (lit.)
SMILES string
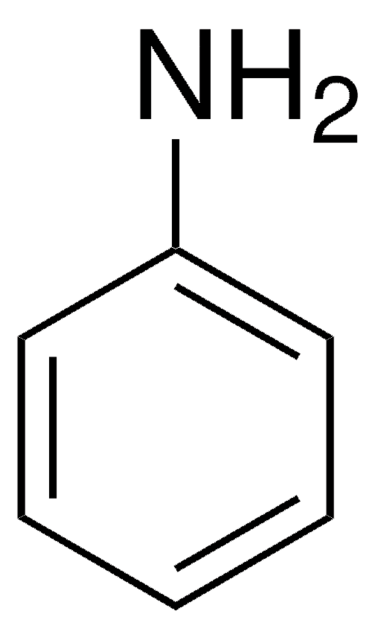
CC(=O)Oc1ccc(C)cc1
InChI
1S/C9H10O2/c1-7-3-5-9(6-4-7)11-8(2)10/h3-6H,1-2H3
InChI key
CDJJKTLOZJAGIZ-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
signalword
Warning
hcodes
Hazard Classifications
Acute Tox. 4 Oral
wgk_germany
WGK 2
flash_point_f
194.0 °F
flash_point_c
90 °C
ppe
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Articles
The Fries rearrangement reaction is an organic name reaction which involves the conversion of phenolic esters into hydroxyaryl ketones on heating in the presence of a catalyst. Suitable catalysts for this reaction are Brønsted or Lewis acids such as HF, AlCl3, BF3, TiCl4, or SnCl4. The Fries rearrangement reaction is an ortho, para-selective reaction, and is used in the preparation of acyl phenols. This organic reaction has been named after German chemist Karl Theophil Fries.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service