655422
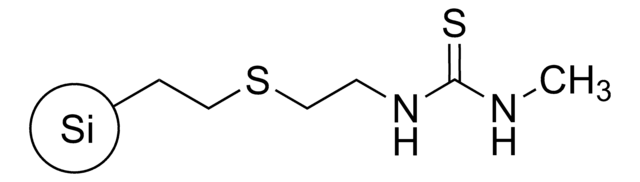
QuadraPure® TU
macroporous, 400-600 μm particle size
Synonym(s):
QuadraPure® Thiourea
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Recommended Products
Quality Level
reaction suitability
reagent type: catalyst
reagent type: chelator
particle size
400-600 μm
General description
QuadraPure® TU is a thiourea-based metal scavenger resin that can be used to prevent metal contamination that occurs during pharmaceutical or fine chemical processing.
Application
Metal Scavenger: Pd, Pt, Ru, Rh, Au, Ag, Cu, Hg, Pb, Cd, Ni, Co, Fe, V, Zn
QuadraPure® TU has been used to remove color from the solution due to leaching of copper species during the copper(I)-mediated 1,2,3-triazole formation via [3+2] cycloaddition of acetylenic compounds with azides. It has also been used as a modifier for carbon pastes to develop QuadraPure® TU residue functionalized resin-modified carbon paste electrode (“TUR-CPE”) for the determination of Pb(II) ions.
Other applications include the removal of metal ions like copper and palladium that leach out during continuous flow coupling reactions.
Other applications include the removal of metal ions like copper and palladium that leach out during continuous flow coupling reactions.
Legal Information
QuadraPure is a registered trademark of Johnson Matthey Finland Oy
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
[3+ 2] Cycloaddition of acetylenes with azides to give 1, 4-disubstituted 1, 2, 3-triazoles in a modular flow reactor.
Smith CD, et al.
Organic & Biomolecular Chemistry, 5(10), 1559-1561 (2007)
Nikzad Nikbin, et al.
Organic Process Research & Development, 11, 458-462 (2007)
Paolo Tosatti et al.
The Journal of organic chemistry, 76(13), 5495-5501 (2011-05-14)
The sequential use of Cu-catalyzed asymmetric allylic alkylation, olefin cross-metathesis, and Ir-catalyzed asymmetric allylic amination allows the concise, stereodivergent synthesis of complex chiral amines with complete regiocontrol and good diastereoselectivity, exemplified by the synthesis of a pair of diastereoisomeric unnatural
Functionalised resin-modified carbon paste sensor for the voltammetric determination of Pb (II) within a wide concentration range.
Mikysek T, et al.
Electrochemical Communications, 10(2), 242-245 (2008)
Michael J. Girgis, et al.
Organic Process Research & Development, 12, 1209-1217 (2008)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service