693243
(S)-Ru(OAc)2(SEGPHOS®)
Synonym(s):
Diacetato[(S)-(−)-5,5′-bis(diphenylphosphino)-4,4′-bi-1,3-benzodioxole]ruthenium(II)
About This Item
Recommended Products
form
powder
Quality Level
storage temp.
2-8°C
InChI
1S/C38H28O4P2.2C2H4O2.Ru/c1-5-13-27(14-6-1)43(28-15-7-2-8-16-28)33-23-21-31-37(41-25-39-31)35(33)36-34(24-22-32-38(36)42-26-40-32)44(29-17-9-3-10-18-29)30-19-11-4-12-20-30;2*1-2(3)4;/h1-24H,25-26H2;2*1H3,(H,3,4);/q;;;+2/p-2
InChI key
BHGLLIGZFQVMBJ-UHFFFAOYSA-L
Related Categories
Application
- To prepare highly chemo, enantio, and diastereoselective primary β-amino lactams by asymmetric reductive amination of racemic β-keto lactams.
- To synthesize chiral primary diarylmethylamines and sterically bulky benzylamines from diaryl and sterically hindered ketones via asymmetric reductive amination reaction.
- For the conversion of levulinic acid to optically active γ-valerolactone via asymmetric hydrogenation reaction.
Legal Information
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Articles
Hydrogenation, Asymmetric Catalysis, Binap, SEGPHOS®, Aldol reaction, Alkenylation, Arylation, Mannich reaction, Fluorination, Michael addition, Hydrosilylation, Cycloaddition, Takasago
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service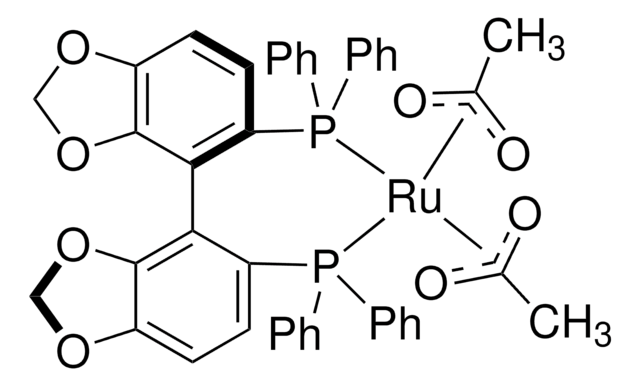
![RuCl2[(R)−DM−BINAP][(R)−DAIPEN]](/deepweb/assets/sigmaaldrich/product/structures/355/628/d82e5e5d-23fd-468a-87ca-db4c45677898/640/d82e5e5d-23fd-468a-87ca-db4c45677898.png)
![Ru(OAc)2[(S)-dtbm-SEGPHOS®] Takasago](/deepweb/assets/sigmaaldrich/product/structures/152/221/1d0db877-5b93-4dc3-af98-eb596623777a/640/1d0db877-5b93-4dc3-af98-eb596623777a.png)
![(R)-RuCl[(p-cymene)(BINAP)]Cl](/deepweb/assets/sigmaaldrich/product/structures/244/078/7a0bdab6-11cc-4030-bbe9-4f687a6a925a/640/7a0bdab6-11cc-4030-bbe9-4f687a6a925a.png)
![RuCl2[(R)−DM−BINAP][(R,R)−DPEN]](/deepweb/assets/sigmaaldrich/product/structures/425/564/80b6a39d-c641-4583-8b52-ab506b343228/640/80b6a39d-c641-4583-8b52-ab506b343228.png)
![(R)-[(RuCl(SEGPHOS®))2(μ-Cl)3][NH2Me2]](/deepweb/assets/sigmaaldrich/product/structures/690/323/6e59fefa-87f1-40b6-b304-70101b15972b/640/6e59fefa-87f1-40b6-b304-70101b15972b.png)
![RuCl2[(S)-(DM-SEGPHOS®)][(S)-DAIPEN]](/deepweb/assets/sigmaaldrich/product/structures/316/926/466dd730-af1f-46d0-a2ac-ed7c8b2abce7/640/466dd730-af1f-46d0-a2ac-ed7c8b2abce7.png)
nickel(II) chloride](/deepweb/assets/sigmaaldrich/product/structures/252/197/3c560697-beb3-4c52-85d6-ebc3af13db69/640/3c560697-beb3-4c52-85d6-ebc3af13db69.png)