All Photos(1)
About This Item
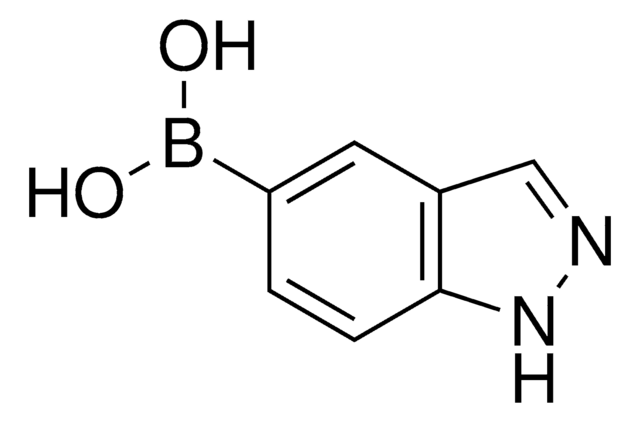
Empirical Formula (Hill Notation):
C7H7BN2O2
CAS Number:
Molecular Weight:
161.95
MDL number:
UNSPSC Code:
12352103
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
form
solid
Quality Level
mp
112-117 °C
storage temp.
2-8°C
SMILES string
OB(O)c1ccc2cn[nH]c2c1
InChI
1S/C7H7BN2O2/c11-8(12)6-2-1-5-4-9-10-7(5)3-6/h1-4,11-12H,(H,9,10)
InChI key
ZKNLCHWRWRYPGG-UHFFFAOYSA-N
Related Categories
Application
Indazole-6-boronic acid can be used as a reactant:
- In Suzuki-Miyaura coupling reaction to synthesize indazole derivatives via C-C bond formation by reacting with different aryl halides.
- To synthesize indazole substituted purines and pyrrolo[2,3-d]pyrimidines as potential kinase inhibitors.
Reactant for preparation of:
- Bicyclic hydroxyphenylmethanone derivatives as hydroxysteroid dehydrogenase inhibitors
- Bisphosphonate inhibitors of human farnesyl pyrophosphate synthase
- Indazolyl benzoimidazoles as PKC-ζ inhibitors
- Pyrazolopyrimidinamine derivatives and their tyrosine and phosphinositide kinase inhibitory activity
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service
![Benzo[c][1,2,5]oxadiazole-5-boronic acid pinacol ester 97%](/deepweb/assets/sigmaaldrich/product/structures/143/941/5b091bed-2dcd-4ac5-b86d-70df392aabce/640/5b091bed-2dcd-4ac5-b86d-70df392aabce.png)