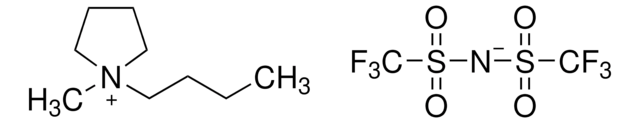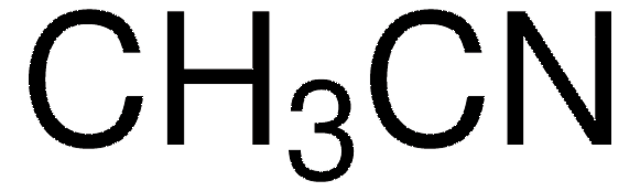900804
1-Butyl-2,3-dimethylimidazolium bis(trifluoromethylsulfonyl)imide
≥99%, H2O <500 ppm
Synonym(s):
1-Butyl-2,3-dimethylimidazolium bis(trifluoromethylsulfonyl)amide, 1-Butyl-2,3-dimethylimidazolium bistriflamide
About This Item
Recommended Products
Quality Level
assay
≥99%
form
liquid
composition
H2O, <500 ppm
impurities
≤500 ppm H2O
bp
430 °C (decomp(lit.))
mp
-76 °C (lit.)
density
1.4059 g/cm3
application(s)
battery manufacturing
InChI
1S/C9H17N2.C2F6NO4S2/c1-4-5-6-11-8-7-10(3)9(11)2;3-1(4,5)14(10,11)9-15(12,13)2(6,7)8/h7-8H,4-6H2,1-3H3;/q+1;-1
InChI key
UCCKRVYTJPMHRO-UHFFFAOYSA-N
Related Categories
Application
related product
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 3 Oral - Eye Dam. 1
wgk_germany
WGK 3
flash_point_f
230.0 °F
flash_point_c
110 °C
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Dr. Sun reviews the recent advances in solid-state rechargeable batteries and cover the fundamentals of solid electrolytes in solid-state batteries, the theory of ion conduction, and the structures and electrochemical processes of solid-state Li batteries.
Here, we present a short review of ionic liquid electrolytes used in state-of-the-art rechargeable batteries including high performance and low-cost aluminum batteries, non-flammable Li-based batteries, and high-cycling and stable dual-graphite batteries. We also outline the key issues explored so as to identify the future direction of IL development.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service