All Photos(1)
About This Item
Linear Formula:
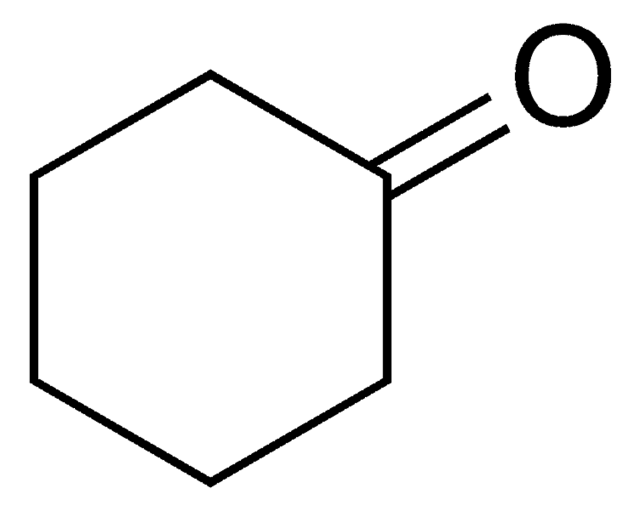
C6H10(=NOH)
CAS Number:
Molecular Weight:
113.16
Beilstein/REAXYS Number:
1616769
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
assay
97%
form
crystals
bp
206-210 °C (lit.)
mp
86-89 °C (lit.)
SMILES string
O\N=C1/CCCCC1
InChI
1S/C6H11NO/c8-7-6-4-2-1-3-5-6/h8H,1-5H2
InChI key
VEZUQRBDRNJBJY-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
signalword
Warning
hcodes
pcodes
Hazard Classifications
Acute Tox. 4 Oral
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Raphaël Turgis et al.
ChemSusChem, 3(12), 1403-1408 (2010-12-01)
Brønsted-acidic ionic liquids that bear a sulfonic acid group, known as Forbes acids, show a good catalytic activity for the Beckmann rearrangement, used to prepare ε-caprolactam, which is a precursor of Nylon 6. The activity essentially stems from the acidity of
D Parmar et al.
Drug metabolism and disposition: the biological fate of chemicals, 19(6), 1101-1107 (1991-11-01)
Cyclohexanone oxime (CHOX), an intermediate used in the synthesis of Polycaprolactam/Nylon, was found to be rapidly absorbed and cleared from the body within 24 hours after a single oral administration of 1, 10 and 30 mg/kg of [14C]-CHOX to the
M J Prival
Mutation research, 497(1-2), 1-9 (2001-08-30)
The basis for the observed mutagenicity of cyclohexanone oxime in the presence of hamster liver S9 in Salmonella typhimurium strain TA1535, but not in TA100, was explored. While the chemical had no effect on the appearance of the background lawn
Wen-Cui Li et al.
Journal of the American Chemical Society, 127(36), 12595-12600 (2005-09-08)
In this study, we present a synthetic pathway for the fabrication of self-supporting zeolite monoliths consisting of crystallized nanoparticles. A resorcinol-formaldehyde-based organic aerogel is used as a template, and silicalite-1 is used as the zeolite example. The silicalite-1 monoliths obtained
S C Gad et al.
Fundamental and applied toxicology : official journal of the Society of Toxicology, 5(1), 128-136 (1985-02-01)
Dermal exposure of rabbits to cyclohexanone oxime (CHO) for 24 hr at 0, 0.8, 2, and 5 g/kg caused dose-related reticulocytosis on the day after dosing as well as a decrease in hemoglobin in the 5-g/kg females 7 days postdosing.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service