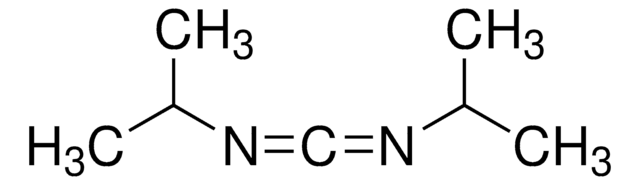D125407
DIC
99%, for peptide synthesis
Synonym(s):
N,N′-Diisopropylcarbodiimide
About This Item
Recommended Products
product name
DIC, 99%
Quality Level
assay
99%
form
liquid
reaction suitability
reaction type: Coupling Reactions
refractive index
n20/D 1.433 (lit.)
bp
145-148 °C (lit.)
density
0.815 g/mL at 20 °C (lit.)
application(s)
peptide synthesis
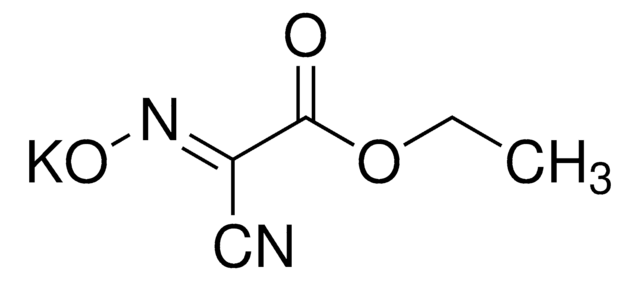
SMILES string
CC(C)N=C=NC(C)C
InChI
1S/C7H14N2/c1-6(2)8-5-9-7(3)4/h6-7H,1-4H3
InChI key
BDNKZNFMNDZQMI-UHFFFAOYSA-N
Gene Information
human ... EPHX2(2053)
mouse ... Ephx2(13850)
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
related product
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 1 Inhalation - Eye Dam. 1 - Flam. Liq. 3 - Resp. Sens. 1 - Skin Sens. 1
wgk_germany
WGK 3
flash_point_f
91.4 °F
flash_point_c
33 °C
ppe
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Amide bonds are ubiquitous in both nature and industrial applications. They are vital to the structure and function of biological macromolecules and polymers. The importance of this functionality has resulted in numerous approaches to its formation, ranging from stoichiometric activation of carboxylic acids to more recent advances in catalytic amide bond formation.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service