M68423
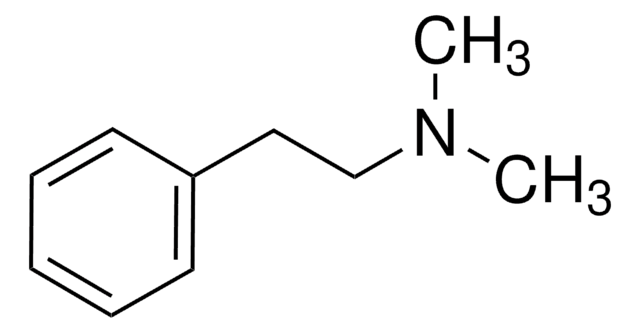
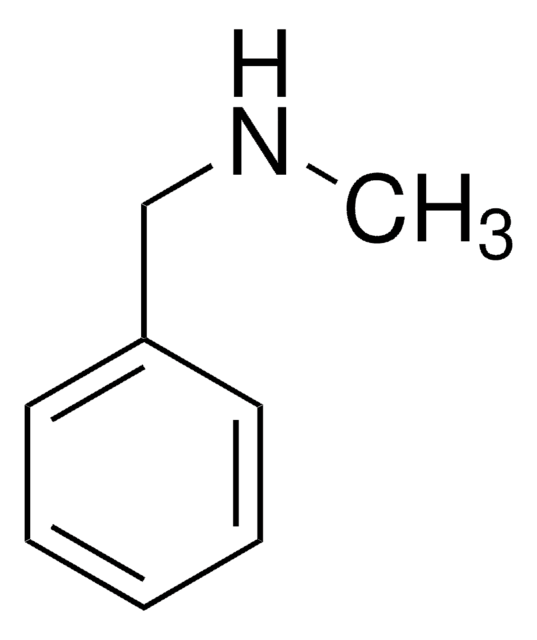
N-Methyl-phenethylamine
99%
Synonym(s):
N-Methyl-2-phenylethylamine
About This Item
Recommended Products
Quality Level
assay
99%
form
liquid
refractive index
n20/D 1.516 (lit.)
bp
203 °C (lit.)
density
0.93 g/mL at 25 °C (lit.)
SMILES string
CNCCc1ccccc1
InChI
1S/C9H13N/c1-10-8-7-9-5-3-2-4-6-9/h2-6,10H,7-8H2,1H3
InChI key
SASNBVQSOZSTPD-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
- To synthesize N-methyl-phenethylamine based tertiary amines by reacting with different alkyl halides in the presence of triphenylphosphine (TPP) and diisopropylazocarboxylate (DIAD) via N-alkylation reaction.
- To fabricate photochemically stable, super-sensitive, and highly selective fluorescent film for the detection of N-methamphetamine (an illicit drug).
- To prepare biologically active squaric acid N-hydroxylamide amide derivatives.
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Eye Dam. 1
Storage Class
10 - Combustible liquids
wgk_germany
WGK 3
ppe
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service