1.07393
Proteinase K
(from Tritirachium album) solution in Tris/HCl pH 7.5; 0.01 mol/l; 600 mAnson-U/ml; for molecular biology EC 3.4.21.14
Synonym(s):
ProK, native proteinase K solution, non-specific protease
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
UNSPSC Code:
12352204
NACRES:
NA.51
Recommended Products
Quality Level
form
liquid
pH
7.5 (25 °C in H2O, undiluted)
density
1.10 g/cm3 at 20 °C
storage temp.
2-8°C
General description
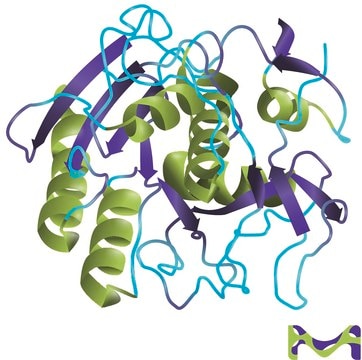
Proteinase K is an endopeptidase that belongs to the subtilisin family of proteinases. The polypeptide chain contains 278 amino acids with the catalytic triad Asp39, His69, Ser224.
Application
Proteinase K has been used:
- to de-crosslink immunoprecipitated samples
- to treat the poly-L-lysine coated slides of colon tissues for terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay
- in in situ hybridization
Biochem/physiol Actions
Proteinase K catalyzes the hydrolysis of esters and peptide bonds. It is used with non-aqueous hydrated solvents for synthesizing peptides. Proteinase K is used to break the cross-linking that develops secondary to formalin fixation and expose the target sequence for primers and polymerase.
Analysis Note
Appearance (colour): colourless
Appearance (clearness): clear
Activity (hemoglobin; pH 7.5; 37 °C): ≥ 600 mAnsonU/ml
Spec. activity (calc. on protein): ≥ 40.0 mAnsonU/mg
DNases (Nicking activity; pBR 322; 6 h; 37 °C): not detectable
RNases (RNA; 2 h; 37°C): not detectable
Colony count (aerobic bacteria): ≤ 10 CFU/ml
Appearance (clearness): clear
Activity (hemoglobin; pH 7.5; 37 °C): ≥ 600 mAnsonU/ml
Spec. activity (calc. on protein): ≥ 40.0 mAnsonU/mg
DNases (Nicking activity; pBR 322; 6 h; 37 °C): not detectable
RNases (RNA; 2 h; 37°C): not detectable
Colony count (aerobic bacteria): ≤ 10 CFU/ml
signalword
Danger
hcodes
pcodes
Hazard Classifications
Resp. Sens. 1
Storage Class
12 - Non Combustible Liquids
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
F Chavagnat et al.
Applied and environmental microbiology, 65(7), 3001-3007 (1999-07-02)
The general aminopeptidase PepN from Streptococcus thermophilus A was purified to protein homogeneity by hydroxyapatite, anion-exchange, and gel filtration chromatographies. The PepN enzyme was estimated to be a monomer of 95 kDa, with maximal activity on N-Lys-7-amino-4-methylcoumarin at pH 7
María Isabel Navarro-Mendoza et al.
Current biology : CB, 29(22), 3791-3802 (2019-11-05)
Centromeres are rapidly evolving across eukaryotes, despite performing a conserved function to ensure high-fidelity chromosome segregation. CENP-A chromatin is a hallmark of a functional centromere in most organisms. Due to its critical role in kinetochore architecture, the loss of CENP-A
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service