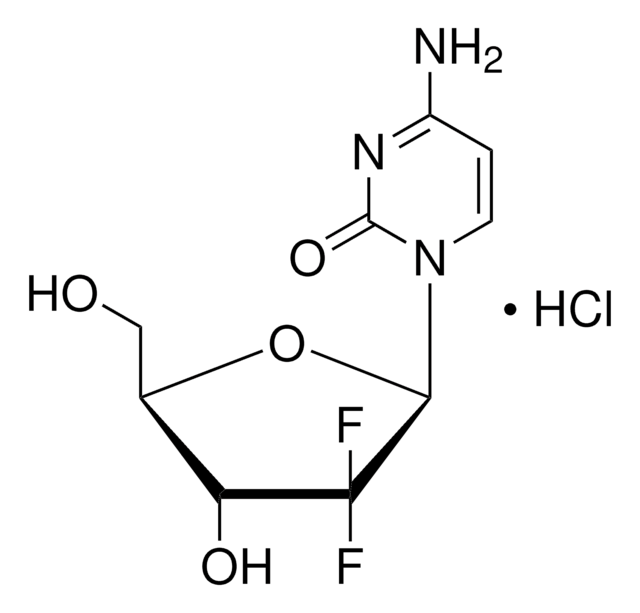
A cell-permeable Atg6/Beclin1-derived autophagy-inducing peptide (Minimum effective conc. ≤10 µM for 3 h) that effectively activates cellular Beclin1 by competing against Beclin1 for binding to its negative regulator GAPR-1/GLIPR2 on the Golgi surface. Shown to induce autosis, autophagy-dependent death, in cells of human, rat, and murine origin in a dose- and time-dependent manner (Minimum effective conc. >5 µM for 5 h in HeLa cultures), blockable by autophagy inhibitor 3-MA (10 mM, Cat. no.189490; [T-B] = 20 µM) or via Na
+/K
+-ATPase inhibition, but insensitive to Z-VAD-FMK (Cat. no.
219007) or Nec-1 (Cat. nos.
480065 &
505224) treatment. Non-cytotoxic autophagy induction with lower doses of Tat-Beclin1 and shorter exposure times is therapeutically beneficial in blocking RNA viral replications both in cultures (4 h 10 µM peptide treatment 4 h post viral infection in HeLa cultures; 0.5-5 µM peptide pre-treatment 24 h before HIV infection in human MDM cultures)
in vitro and in CHIKV-infected neonatal mice (15 mg/kg/day i.p.)
in vivo. While Tat-Beclin1 bioavailability is limited to the peripheral tissues in mice, a peptide based on Tat-Beclin1 retro-inverso sequence (Cat. no. 506416) shows efficacy in a murine model of neonatal WNV CNS infection. Note: increased drug potency is reported in acidified (pH 7.0) serum-free cultures.
A cell-permeable autophagy-inducing peptide (Minimum effective conc. ≤10 µM assessed by increased LC3-II/LC3-I ratio & p62 downregulation post 3 h treatment in A549, COS-7, HBEC30-KT, HCC827, HeLa, MEF, THP-1 cultures) that is composed of essential HIV-1 virulence factor Nef-binding sequence derived from human Atg6/Beclin1 (aa 267-284) evolutionarily conserved domain (ECD) with substitutions at three non-species-conserved residues (H275E, S279D, and Q280E) for enhanced solubility and N-terminally fused to the membrane-permeant HIV-1 Tat protein transduction domain (PTD) sequence (aa 47-57) via a -Gly-Gly- linkage to facilitate cellular delivery and Beclin1 activation via competitive binding to its negative regulator "Golgi-associated plant pathogenesis-related protein-1" (GAPR-1/GLIPR2) on the Golgi surface. Shown to effectively induce autosis, autophagy-dependent death morphologically and pharmacologically distinct from apoptosis and necrosis, in various cell cultures of human, rat, and murine origin in a dose- and time-dependent manner (Minimum effective conc. >5 µM post 5 h treatment as assessed by HeLa Trypan blue staining; Fold of control Trypan blue-positive population/20 µM peptide exposure time = 3/1 h, 9/4 h, 20/8 h), blockable by autophagy inhibitor 3-MA (10 mM, Cat. no. 189490; [T-B] = 20 µM) or via Na+/K+-ATPase inhibition by cardiac glycosides, but insensitive to Z-VAD-FMK (Cat. no. 219007) or Nec-1 (Cat. nos. 480065 & 505224) treatment. Autophagy induction by Tat-Beclin1 treatment is also reported to result in clearance of small (<1 µm) CFP-htt103Q aggregates in HeLa cells (59% and 85% reduction, respectively, of aggregates-positive population and average aggregates per cell; 20 µM/4h/d for 2 d) and non-cytotoxic autophagy induction with lower level of Tat-Beclin1 dosage and limited exposure time is demonstrated to be therapeutically beneficial in blocking RNA viral replications both in cultures (4 h, 10 µM peptide treatment 4 h post CHIKV, SINV, or WNV infection in HeLa cultures; 0.5-5 µM peptide pre-treatment 24 h prior to HIV infection in human MDM cultures) in vitro and in CHIKV-inoculated neonatal mice (15 mg/kg/day i.p.) in vivo. While Tat-Beclin1 bioavailability is limited to the peripheral tissues in mice, a peptide based on Tat-Beclin1 retro-inverso sequence (Cat. no. 506416) shows efficacy in a murine model of neonatal WNV CNS infection. Note: increased drug potency is reported in acidified (pH 7.0) serum-free cultures.