8.51014
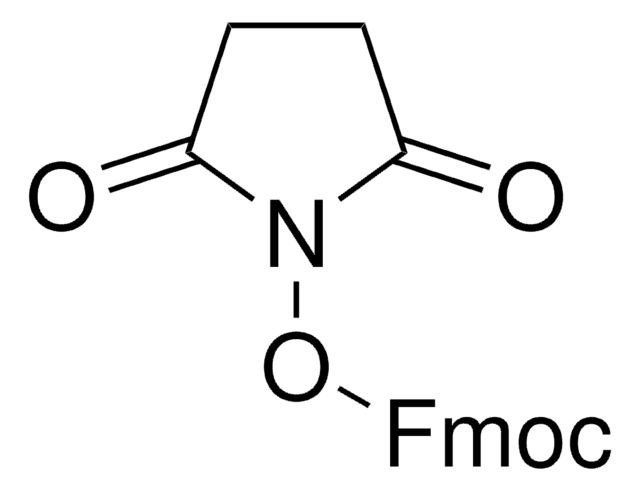
Fmoc-OSu
Novabiochem®
Synonym(s):
Fmoc-OSu, FMOC-ONSu, N-(9H-Fluoren-9-ylmethoxycarbonyloxy)succinimide, N-(9-Fluorenylmethoxycarbonyloxy)succinimide, 9-Fluorenylmethyl-succinimidyl carbonate
About This Item
Recommended Products
Quality Level
product line
Novabiochem®
assay
≥99.0% (HPLC)
form
powder
manufacturer/tradename
Novabiochem®
mp
145-148 °C
application(s)
peptide synthesis
functional group
Fmoc
storage temp.
2-8°C
InChI
1S/C19H15NO5/c21-17-9-10-18(22)20(17)25-19(23)24-11-16-14-7-3-1-5-12(14)13-6-2-4-8-15(13)16/h1-8,16H,9-11H2
InChI key
WMSUFWLPZLCIHP-UHFFFAOYSA-N
Related Categories
General description
Literature references
[1] A. Paquet (1982) Can. J. Chem., 60, 976.
[2] L. Lapatsanis, et al. (1983) Synthesis, 671.
[3] G. F. Sigler, et al. (1983) Biopolymers, 22, 2157.
Application
- Sequence-defined positioning of amine and amide residues to control catechol driven wet adhesion: This study uses Fmoc-OSu for selective functionalization of amines, demonstrating its role in synthesizing complex molecules with specific adhesive properties (Fischer et al., 2020).
- Fmoc-OASUD: A new reagent for the preparation of Fmoc-amino acids free from impurities resulting from Lossen rearrangement: Discusses the development of Fmoc-OASUD as a superior alternative to Fmoc-OSu, reducing impurities in peptide synthesis processes (Rao et al., 2016).
Linkage
Analysis Note
Appearance of substance (visual): powder
Identity (IR): passes test
Assay (HPLC, area%): ≥ 99.0 % (a/a)
Solubility (1 mmole in 2 ml DMF): clearly soluble
Legal Information
signalword
Warning
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Aquatic Chronic 2 - Skin Sens. 1
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service