SGW1A47HH3
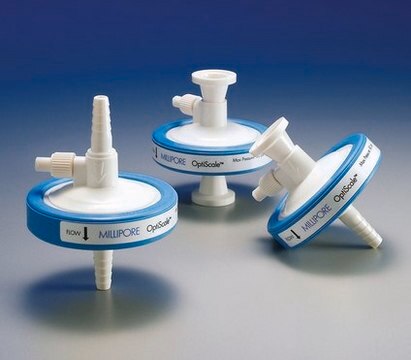
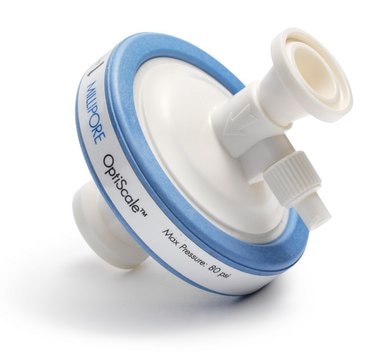
Polysep™ II 1.0/0.2/0.1 µm nominal, OptiScale® Capsule
OptiScale® 47, filtration area 17.7 cm2, pore size 1.0/0.2/0.1 μm
Synonym(s):
Optiscale Capsule Polysep II 1.0/0.2/0.1 μm 1/4 in. HB/HB
About This Item
Recommended Products
material
PVDF vent cap
borosilicate glass fiber (BGF) membrane
fluoroelastomer FKM seal
mixed cellulose esters (MCE) membrane
polycarbonate
polypropylene support
Quality Level
reg. compliance
meets FDA Indirect Food Additive requirements cited in 21 CFR 177-182 (all component materials)
sterility
non-sterile
sterilization compatibility
autoclavable compatible
product line
OptiScale® 47
feature
hydrophilic
manufacturer/tradename
OptiScale®
parameter
25 °C max. inlet temp.
5.5 bar max. inlet pressure (80 psi) at 25 °C
80 psig max. inlet pressure
technique(s)
prefiltration: suitable
diam.
6.9 cm (2.7 in.)
filtration area
17.7 cm2
inlet connection diam.
1/4 in.
outlet connection diam.
1/4 in.
impurities
≤0.25 EU/mL bacterial endotoxins (LAL test, Aqueous extraction)
matrix
Polysep™ II
pore size
1.0/0.2/0.1 μm nominal pore size
1.0/0.2/0.1 μm pore size
input
sample type liquid
fitting
inlet hose barb
outlet hose barb
(6 mm (1/4 in.) Hose Barb Inlet and Outlet)
Related Categories
General description
Features and Benefits
Other Notes
- Organism Retention: Bacterial
- Mode of Action: Filtration (size exclusion)
- Application: Protein purification
- Intended Use: Reduction or removal of microorganism/bioburden
- Instructions for Use: Please refer installation and wetting sections of OPTISCALE®-25 user guide
- Storage Statement: Please refer OPTISCALE®-25 user guide
- Disposal Statement: Dispose of in accordance with applicable federal, state and local regulations.
Preparation Note
3 autoclave cycles of 60 min @ 121 °C
Analysis Note
Legal Information
Still not finding the right product?
Give our Product Selector Tool a try.
signalword
Danger
hcodes
Hazard Classifications
Carc. 1B Inhalation
wgk_germany
WGK 2
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Related Content
A relatively templated manufacturing process can be used to produce live vector vaccines. Challenges exist, however, and include yield loss due to sterile filtration, vector aggregation and scaling adherent cultures. Learn all about this core platform technology and how expertise—empowered by collaboration— can overcome manufacturing challenges.
Manufacturing viral vaccines is complex and there is no templated approach. Each process must be tailored to the shape, size, nature, physico-chemical behavior, stability, and host specificity of the virus. Learn all about this core platform technology and how expertise—empowered by collaboration— can overcome manufacturing challenges.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service