09954
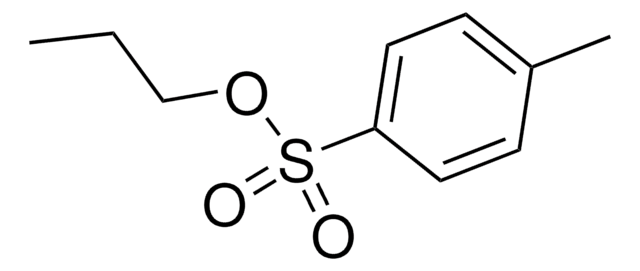
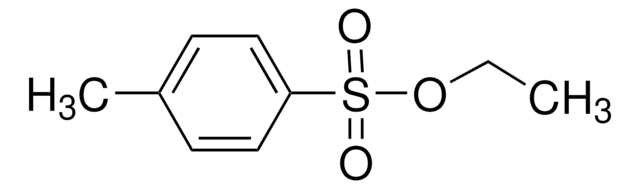
Propargyl p-toluenesulfonate
≥97.0% (GC)
Synonym(s):
Propargyl tosylate
About This Item
Recommended Products
assay
≥97.0% (GC)
refractive index
n20/D 1.530
density
1.215 g/mL at 20 °C (lit.)
storage temp.
2-8°C
SMILES string
Cc1ccc(cc1)S(=O)(=O)OCC#C
InChI
1S/C10H10O3S/c1-3-8-13-14(11,12)10-6-4-9(2)5-7-10/h1,4-7H,8H2,2H3
InChI key
LMBVCSFXFFROTA-UHFFFAOYSA-N
Related Categories
Application
It can also be used as a reagent to synthesize:
- 2-hydroxy-4-pentynoic acid by an alkylation reaction with diethyl 2-acetamidomalonate followed by subsequent hydrolysis, decarboxylation, diazotization, and hydroxylation reactions.
- Furan derivatives by Pd-catalyzed reaction with acylchromates.
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
10 - Combustible liquids
wgk_germany
WGK 3
flash_point_f
212.0 °F - closed cup
flash_point_c
100 °C - closed cup
ppe
Eyeshields, Gloves, type ABEK (EN14387) respirator filter
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Sorry, we don't have COAs for this product available online at this time.
If you need assistance, please contact Customer Support.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service