20025
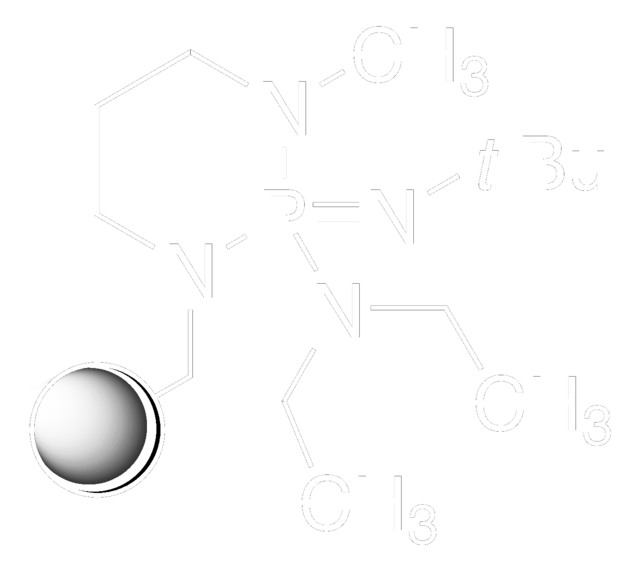
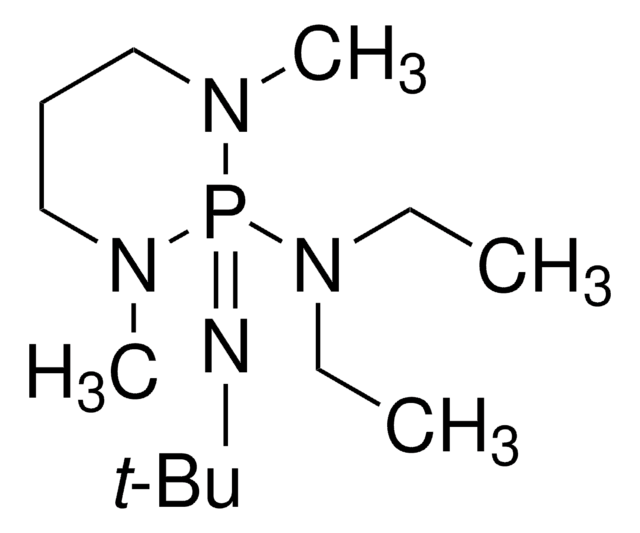
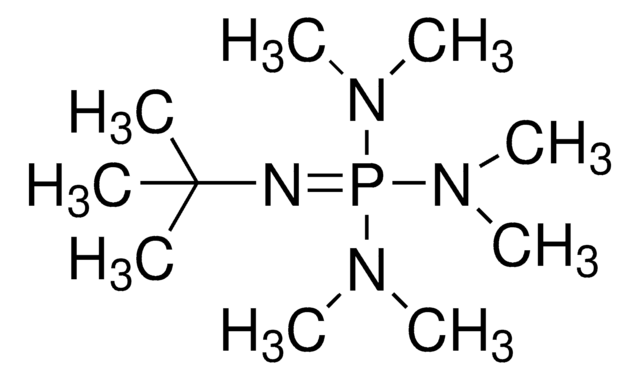
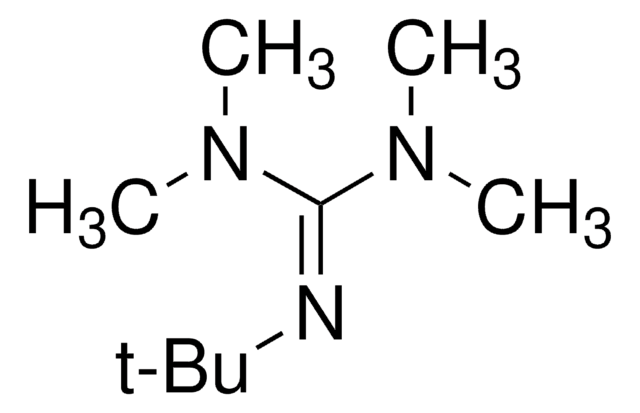
2-tert-Butylimino-2-diethylamino-1,3-dimethylperhydro-1,3,2-diazaphosphorine
purum, ≥98.0% (GC)
Synonym(s):
BEMP
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C13H31N4P
CAS Number:
Molecular Weight:
274.39
Beilstein/REAXYS Number:
5534901
MDL number:
UNSPSC Code:
12352005
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
grade
purum
Quality Level
assay
≥98.0% (GC)
form
liquid
refractive index
n20/D 1.477 (lit.)
n20/D 1.477
bp
74 °C/0.03 mmHg (lit.)
density
0.948 g/mL at 25 °C (lit.)
SMILES string
CCN(CC)P1(=NC(C)(C)C)N(C)CCCN1C
InChI
1S/C13H31N4P/c1-8-17(9-2)18(14-13(3,4)5)15(6)11-10-12-16(18)7/h8-12H2,1-7H3
InChI key
VSCBATMPTLKTOV-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
2-tert-Butylimino-2-diethylamino-1,3-dimethylperhydro-1,3,2-diazaphosphorine (BEMP) is a phosphazene base. BEMP participates in the mild base catalyzed nucleophilic ring opening of N-sulfonyl aziridines. BEMP supported on polystyrene (PS-BEMP) has been reported as an efficient catalyst for the ring-opening of epoxides with phenols. BEMP is about 2000 times more basic and also much more sterically hindered than DBU (l,8-diazabicyclo[5.4.0]undecene).
Application
2-tert-Butylimino-2-diethylamino-1,3-dimethylperhydro-1,3,2-diazaphosphorine (BEMP), a organic phosphorine base may be used in the following studies:
- Synthesis of a 2,3-dihydrobenzo[1,4]dioxepin-5-one.
- As catalyst in the alkylation reactions of carbon acids.
- As organocatalyst in the controlled "immortal" ring-opening polymerization (iROP) of six-membered cyclic carbonates.
signalword
Danger
hcodes
Hazard Classifications
Skin Corr. 1B
wgk_germany
WGK 3
flash_point_f
230.0 °F
flash_point_c
110 °C
ppe
Faceshields, Gloves, Goggles, type ABEK (EN14387) respirator filter
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
2-tert-Butylimino-2-diethylamino-1, 3-dimethylperhydro-1, 3, 2-diazaphosphorine Supported on Polystyrene (PS-BEMP) as an Efficient Recoverable and Reusable Catalyst for the Phenolysis of Epoxides under Solvent-Free Conditions.
Zvagulis A, et al.
Advanced Synthesis & Catalysis, 352(14-15), 2489-2496 (2010)
Ke Wen et al.
The Journal of organic chemistry, 67(22), 7887-7889 (2002-10-26)
A short and efficient synthesis of 2'-O-methoxyethylguanosine (8) is described. Central to this strategy is the development of a novel silicon-based protecting group (MDPSCl(2), 2) used to protect the 3',5'-hydroxyl groups of the ribose. Silylation of guanosine with 2 proceeded
Tommaso Angelini et al.
Organic & biomolecular chemistry, 11(30), 5042-5046 (2013-06-28)
An efficient protocol for the hydrophosphonylation of aromatic and aliphatic aldehydes catalyzed by PS-BEMP under solvent-free conditions (SolFC) has been reported. Addition reactions were performed by using equimolar amounts of reagents and the resulting α-hydroxyphosphonates were isolated with simple workup
Thomas A Moss et al.
Chemical communications (Cambridge, England), (21)(21), 2474-2476 (2008-05-21)
N-Mesitylene sulfonyl and N-tosyl aziridines have been identified as effective electrophiles in alkylation reactions of carbon acids catalyzed by the organic phosphorine base BEMP; yields of up to 99% for a range of pro-nucleophiles under mild reaction conditions are reported.
M.J. O'Donnell et al.
Tetrahedron Letters, 39, 8775-8775 (1998)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service
![7-Methyl-1,5,7-triazabicyclo[4.4.0]dec-5-ene 98%](/deepweb/assets/sigmaaldrich/product/structures/237/769/028967ef-ca63-4f22-acc9-68f135a43b9a/640/028967ef-ca63-4f22-acc9-68f135a43b9a.png)
![1,5,7-Triazabicyclo[4.4.0]dec-5-ene 98%](/deepweb/assets/sigmaaldrich/product/structures/171/446/333d560c-cff6-4958-b489-5acfb3057cce/640/333d560c-cff6-4958-b489-5acfb3057cce.png)