247588
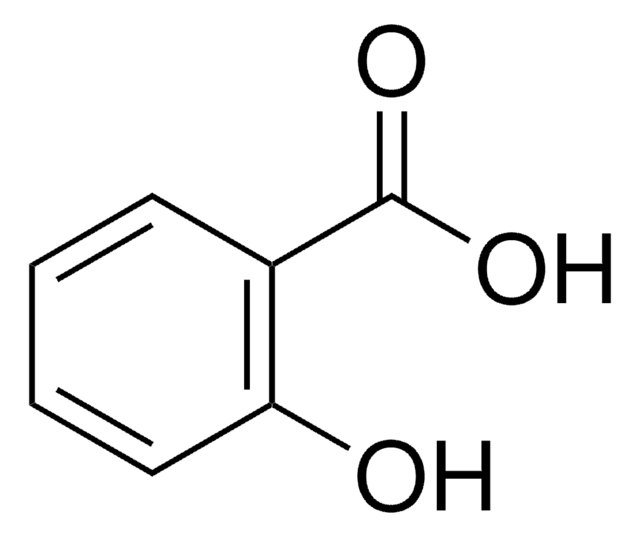
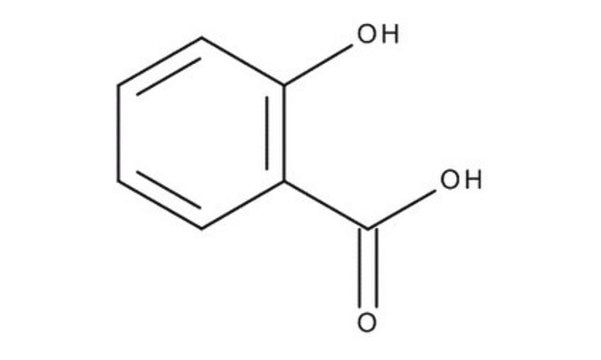
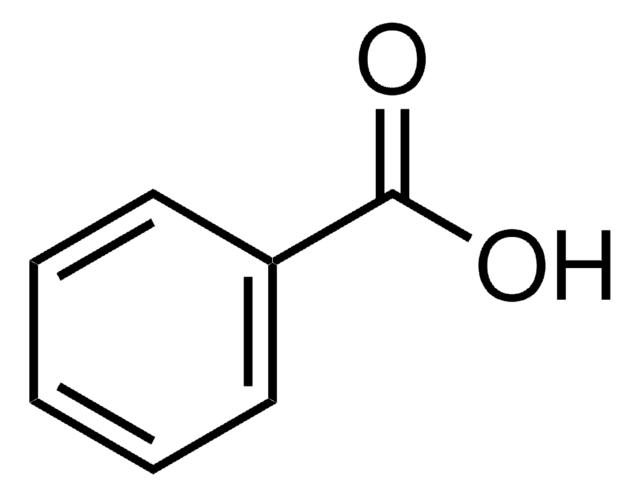
Salicylic acid
ACS reagent, ≥99.0%
Synonym(s):
2-Hydroxybenzoic acid
About This Item
Recommended Products
grade
ACS reagent
Quality Level
vapor density
4.8 (vs air)
vapor pressure
1 mmHg ( 114 °C)
assay
≥99.0%
impurities
H2SO4, passes test (darkened)
ign. residue
≤0.01%
bp
211 °C (lit.)
mp
158-161 °C (lit.)
anion traces
chloride (Cl-): ≤0.001%
sulfate (SO42-): ≤0.003%
cation traces
Fe: ≤2 ppm
heavy metals (as Pb): ≤5 ppm
SMILES string
OC(=O)c1ccccc1O
InChI
1S/C7H6O3/c8-6-4-2-1-3-5(6)7(9)10/h1-4,8H,(H,9,10)
InChI key
YGSDEFSMJLZEOE-UHFFFAOYSA-N
Gene Information
human ... ALB(213) , PTPN1(5770)
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
- as one of the chemical compound in exposure-based validation of in vitro gastrulation model employed in developmental toxicity screening assays
- as a reference standard in salicylate quantification by reverse-phase high-performance liquid chromatography (RP-HPLC)
Biochem/physiol Actions
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Eye Dam. 1 - Repr. 2
Storage Class
11 - Combustible Solids
wgk_germany
WGK 1
flash_point_f
314.6 °F - closed cup
flash_point_c
157 °C - closed cup
ppe
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Sorry, we don't have COAs for this product available online at this time.
If you need assistance, please contact Customer Support.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service