31550
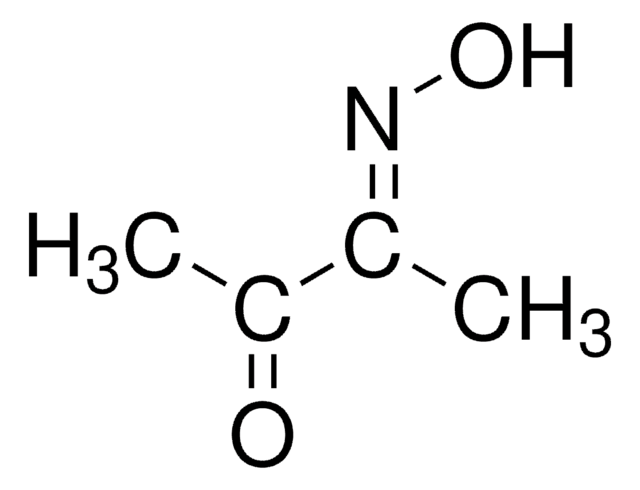
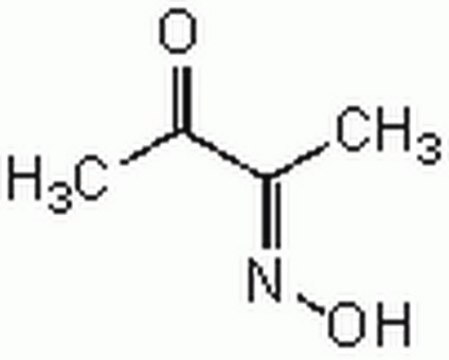
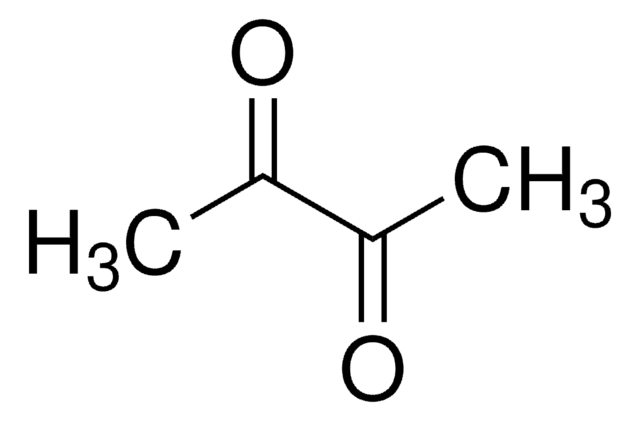
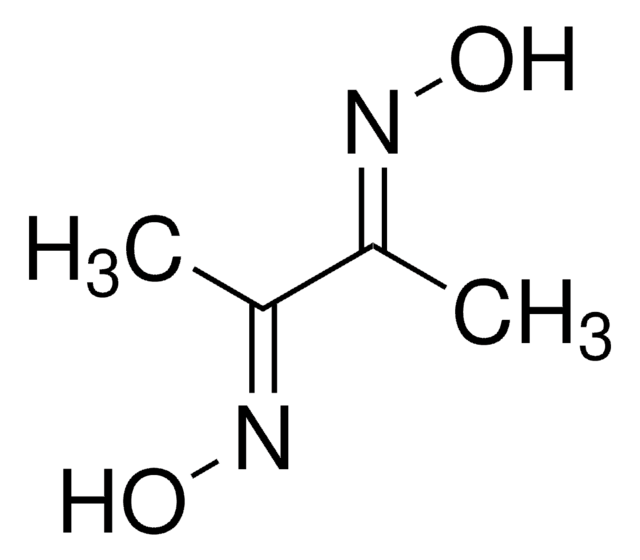
2,3-Butanedione monoxime
for spectrophotometric det. of urea, ≥99.0%
Synonym(s):
BDM, Biacetyl monoxime, Diacetyl monoxime
About This Item
Recommended Products
Quality Level
assay
≥98.0% (N)
≥99.0%
form
solid
quality
for spectrophotometric det. of urea
technique(s)
UV/Vis spectroscopy: suitable
ign. residue
≤0.05% (as SO4)
bp
185-186 °C (lit.)
mp
75-76 °C
75-78 °C (lit.)
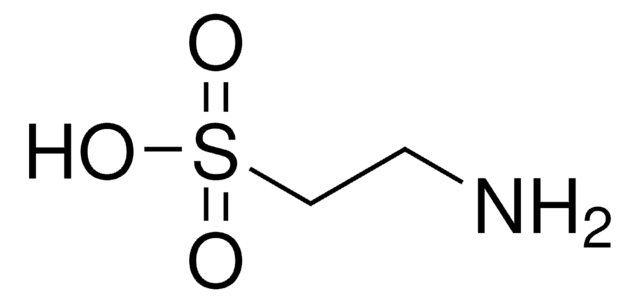
SMILES string
CC(=O)\C(C)=N\O
InChI
1S/C4H7NO2/c1-3(5-7)4(2)6/h7H,1-2H3/b5-3+
InChI key
FSEUPUDHEBLWJY-HWKANZROSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
- A novel method to extend viability and functionality of living heart slices.: This research introduces a novel application of 2,3-Butanedione monoxime for prolonging the functional lifespan of cardiac tissue samples in experimental settings, offering insights into cardiac biology and potential therapeutic targets (Ross et al., 2023).
- Molecular Mechanisms of Deregulation of Muscle Contractility Caused by the R168H Mutation in TPM3 and Its Attenuation by Therapeutic Agents.: The study utilizes 2,3-Butanedione monoxime to investigate the molecular pathways affected by genetic mutations in muscle contractility, contributing to the understanding of muscle disorders and their management (Karpicheva et al., 2023).
- Generation of myocyte agonal Ca(2+) waves and contraction bands in perfused rat hearts following irreversible membrane permeabilisation.: Research employing 2,3-Butanedione monoxime investigates its role in inducing specific cellular events in cardiac cells under stress, highlighting its potential in studies of heart disease mechanisms and therapies (Morishita et al., 2023).
Analysis Note
0.5 g are completely soluble and give a clear solution in 10 mL water or also in 10 mL ethanol.
Sensitivity test:
0.05 mg urea in 3 mL water and 5 mL conc. HCl together with a 3% solution in water of diacetylmonoxime heated on a water bath for 10 minutes give a light yellow color.
Other Notes
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Sorry, we don't have COAs for this product available online at this time.
If you need assistance, please contact Customer Support.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service