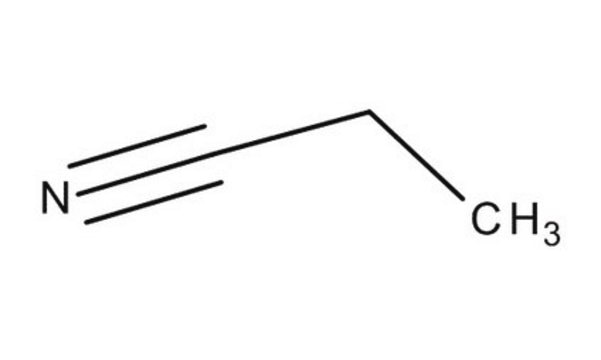
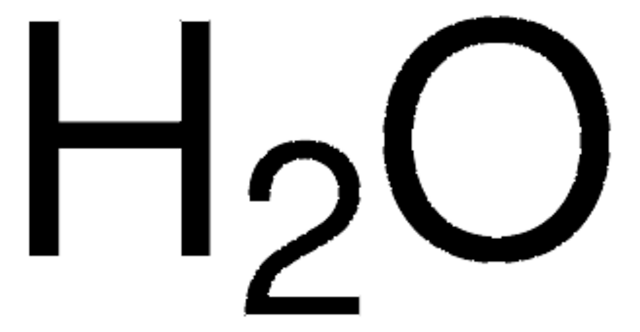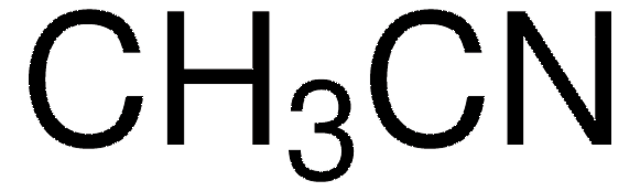81900
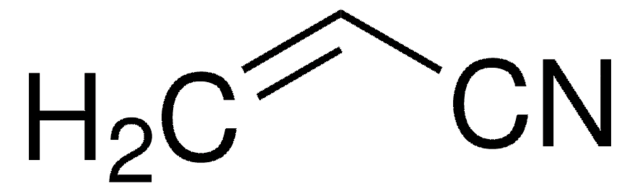
Propionitrile
purum, ≥99.0% (GC)
Synonym(s):
PPN, Ethyl cyanide
About This Item
Recommended Products
grade
purum
Quality Level
assay
≥99.0% (GC)
form
liquid
refractive index
n20/D 1.366 (lit.)
n20/D 1.366
bp
97 °C (lit.)
mp
−93 °C (lit.)
density
0.772 g/mL at 25 °C (lit.)
SMILES string
CCC#N
InChI
1S/C3H5N/c1-2-3-4/h2H2,1H3
InChI key
FVSKHRXBFJPNKK-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
- Propionitrile (PPN) is an effective solvent for catalytic asymmetric aldol reaction of a silyl enol ether with aldehydes in the presence of a chiral tin(II) Lewis acid catalyst.
- The co-solvent formed by mixing PPN with acetonitrile can be used to fabricate polymer gel electrolytes (PGEs) of dye-sensitized solar cells (DSSCs), which lead to enhanced stability of gel-state DSSCs.
- PPN can be used as a solvent for the Brønsted acid-catalyzed synthesis of N-alkyl cis-aziridines via [2+1] annulation of a diazo compound formed by the combination of an acetate and enolate. The process does not involve the use of metals or reagents and only atomic nitrogen as a co-product.
signalword
Danger
Hazard Classifications
Acute Tox. 2 Dermal - Acute Tox. 2 Oral - Acute Tox. 4 Inhalation - Eye Irrit. 2 - Flam. Liq. 2
wgk_germany
WGK 1
flash_point_f
42.8 °F
flash_point_c
6 °C
ppe
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Sorry, we don't have COAs for this product available online at this time.
If you need assistance, please contact Customer Support.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service