242772
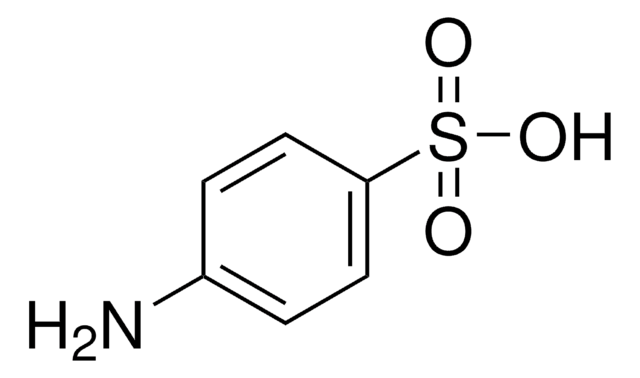
Sulfamic acid
ReagentPlus®, ≥99%
Synonym(s):
Amidosulfonic acid
About This Item
Quality Level
product line
ReagentPlus®
assay
≥99%
form
powder or crystals
mp
215-225 °C (dec.) (lit.)
solubility
water: 50 mg/mL, clear, colorless
density
2.151 g/cm3 at 25 °C
SMILES string
NS(O)(=O)=O
InChI
1S/H3NO3S/c1-5(2,3)4/h(H3,1,2,3,4)
InChI key
IIACRCGMVDHOTQ-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
The ability of sulfamic acid/zinc powder to reduce nitrate to nitrogen gas under acidic condition has been utilized to denitrify nitrate-rich wastewater.
Application
- As a catalyst in the synthesis of aryl-14H-dibenzo[a.j]xanthenes.
- As a green catalyst for the liquid Beckmann rearrangement of ketoxime in dried acetonitrile.
- As a nitrogen source as well as an in situ activator of hydroxy groups in the direct iridium-catalyzed synthesis of primary allylic amines from allylic alcohols.
- Synthesis of polysubstituted quinolones.
Legal Information
signalword
Warning
hcodes
Hazard Classifications
Aquatic Chronic 3 - Eye Irrit. 2 - Skin Irrit. 2
Storage Class
8B - Non-combustible, corrosive hazardous materials
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service