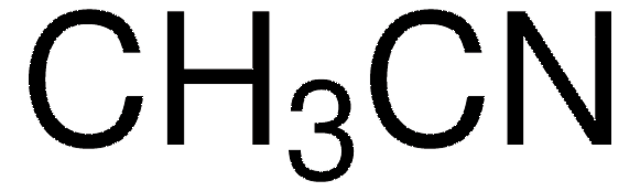439134
Acetonitrile
≥99.9%, gradient grade, suitable for UV/Vis spectroscopy, HPLC
Synonym(s):
ACN, Cyanomethane, Ethyl nitrile, Methyl cyanide
About This Item
product name
Acetonitrile, suitable for HPLC, gradient grade, ≥99.9%
grade
gradient grade
Quality Level
vapor density
1.41 (vs air)
vapor pressure
72.8 mmHg ( 20 °C)
description
HPLC gradient analysis <= 0.5 mAU @254 nm
HPLC gradient analysis <= 3 mAU @ 210 nm
baseline drift at 210 nm <= 15 mAU
assay
≥99.9%
form
liquid
autoignition temp.
973 °F
purified by
sub-micron filtration
expl. lim.
16 %
technique(s)
HPLC: suitable
UV/Vis spectroscopy: suitable
impurities
≤0.0002% free alkali (as NH3)
≤0.001% free acid (as CH3COOH)
≤0.01% water (Karl Fischer)
≤0.5 ppb quinine fluor., 365 nm
≤1 ppb quinine fluor., 254 nm
evapn. residue
<0.0002%
refractive index
n20/D 1.344 (lit.)
bp
81-82 °C (lit.)
mp
−45 °C (lit.)
solubility
water: soluble
density
0.786 g/mL at 25 °C (lit.)
λ
H2O reference
UV absorption
λ: 195 nm Amax: ≤0.12
λ: 200 nm Amax: ≤0.032
λ: 230 nm Amax: ≤0.0044
application(s)
food and beverages
format
neat
SMILES string
CC#N
InChI
1S/C2H3N/c1-2-3/h1H3
InChI key
WEVYAHXRMPXWCK-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
- Chromatographic Separation of Anthocyanins: Acetonitrile is a preferred solvent in high-performance liquid chromatography (HPLC) for the separation of anthocyanins and other phenolic compounds. Studies have explored the selectivity control of anthocyanin separation by replacing acetonitrile with methanol in the mobile phase, highlighting the solvent′s role in optimizing chromatographic conditions for improved resolution and detection sensitivity (Deineka et al., Journal of Analytical Chemistry, 2021).
- Peroxyoxalate Chemiluminescence Detection: Acetonitrile is used in peroxyoxalate chemiluminescence for the detection of α-amino acids. The solvent′s ability to form stable mixed solutions with water and ethyl acetate enhances the chemiluminescence reaction, allowing for sensitive and selective detection of amino acids in various samples. This application is essential for biochemical and clinical analyses (Kan et al., American Journal of Analytical Chemistry, 2020).
- Electrochemical Detection of DNA Modifications: Acetonitrile is utilized in electrochemical methods for detecting DNA modifications, such as 5-formyluracil. The solvent′s role in stabilizing the (2-benzimidazolyl) acetonitrile labeling reaction enhances the selectivity and sensitivity of detecting DNA modifications, which is crucial for genomic and biomedical research (Tang et al., Analytical Chemistry, 2021).
- Alternative Solvent in Chromatography: Research has investigated replacing acetonitrile with ethanol in reversed-phase HPLC for determining anthocyanins. This substitution aims to maintain chromatographic performance while reducing the environmental impact and costs associated with acetonitrile use. The findings demonstrate the feasibility of using ethanol as a greener alternative without compromising analytical quality (Deineka et al., Journal of Analytical Chemistry, 2023).
Preparation Note
related product
signalword
Danger
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Eye Irrit. 2 - Flam. Liq. 2
Storage Class
3 - Flammable liquids
wgk_germany
WGK 2
flash_point_f
35.6 °F - closed cup
flash_point_c
2.0 °C - closed cup
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service