D8174
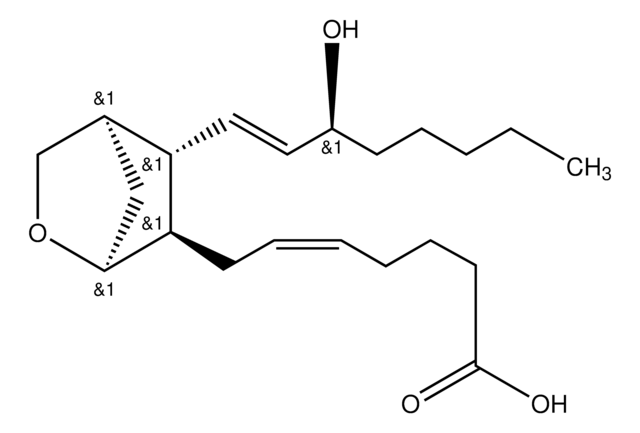
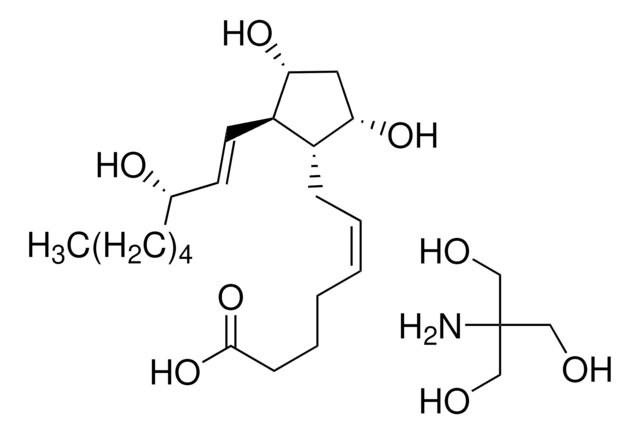
9,11-Dideoxy-11α,9α-epoxymethanoprostaglandin F2α
solution, 10 mg/mL in methyl acetate
Synonym(s):
U-46619, U46619
About This Item
Recommended Products
form
solution
Quality Level
concentration
10 mg/mL in methyl acetate
shipped in
dry ice
storage temp.
−20°C
SMILES string
CCCCC[C@H](O)\C=C\[C@H]1C2CC(CO2)[C@@H]1C\C=C/CCCC(O)=O
InChI
1S/C21H34O4/c1-2-3-6-9-17(22)12-13-19-18(16-14-20(19)25-15-16)10-7-4-5-8-11-21(23)24/h4,7,12-13,16-20,22H,2-3,5-6,8-11,14-15H2,1H3,(H,23,24)/b7-4-,13-12+/t16?,17-,18-,19+,20?/m0/s1
InChI key
LQANGKSBLPMBTJ-REGKDVDGSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- to induce aortic smooth muscle (SM) contraction in mice deficient in myosin light chain 9 (Myl9) gene
- to induce contraction as part of vascular reactivity experiments using mice aorta
- as a thromboxane/prostaglandin agonist to study the effect of dithiothreitol (DTT) on mice arterial vessel viability
Biochem/physiol Actions
Features and Benefits
signalword
Danger
hcodes
Hazard Classifications
Eye Irrit. 2 - Flam. Liq. 2 - STOT SE 3
target_organs
Central nervous system
supp_hazards
Storage Class
3 - Flammable liquids
wgk_germany
WGK 2
flash_point_f
14.0 °F
flash_point_c
-10 °C
ppe
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Related Content
Discover Bioactive Small Molecules for Lipid Signaling Research
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service