K1751
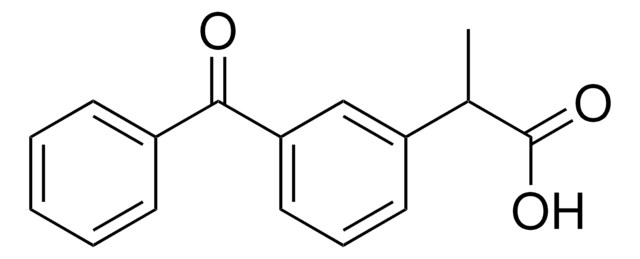
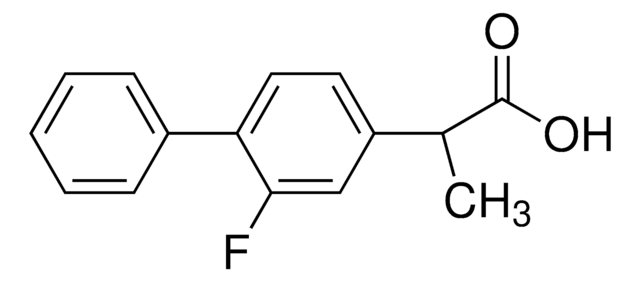
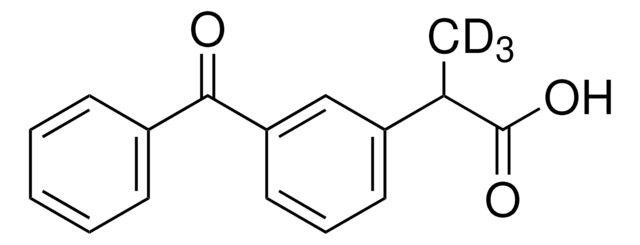
Ketoprofen
≥98% (TLC)
Synonym(s):
2-(3-Benzoylphenyl)propionic acid
About This Item
Recommended Products
biological source
synthetic
Quality Level
assay
≥98% (TLC)
form
powder
solubility
ethanol: 50 mg/mL, clear, colorless to yellow
originator
Bayer
SMILES string
CC(C(O)=O)c1cccc(c1)C(=O)c2ccccc2
InChI
1S/C16H14O3/c1-11(16(18)19)13-8-5-9-14(10-13)15(17)12-6-3-2-4-7-12/h2-11H,1H3,(H,18,19)
InChI key
DKYWVDODHFEZIM-UHFFFAOYSA-N
Gene Information
human ... ALB(213) , IL8RA(3577) , PTGS1(5742) , PTGS2(5743)
Looking for similar products? Visit Product Comparison Guide
General description
Application
- as sample to analyse the effects of long storage period by chromatographic and microscopic techniques
- as a nonselective COX inhibitor to inject subcutaneously in rats to study its effect on stress/methamphetamine hydrochloride-induced changes in occludin, claudin-5 and COX-2 protein immunoreactivity, truncation of β-dystroglycan, brain water content and fluorescein isothiocyanate-dextran extravasation
- in phosphate buffer to study its ability to inhibit heat-induced denaturation of albumin
Biochem/physiol Actions
Features and Benefits
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 3 Oral
Storage Class
6.1C - Combustible, acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
wgk_germany
WGK 2
ppe
Eyeshields, Faceshields, Gloves, type P2 (EN 143) respirator cartridges
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Human epithelial intestinal colonic organoids can be used as an alternative to Caco-2 drug permeability assays for drug screening and compound toxicity testing.
Related Content
Discover Bioactive Small Molecules for Lipid Signaling Research
Chromatograms
application for HPLCapplication for HPLCapplication for HPLCOur team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service