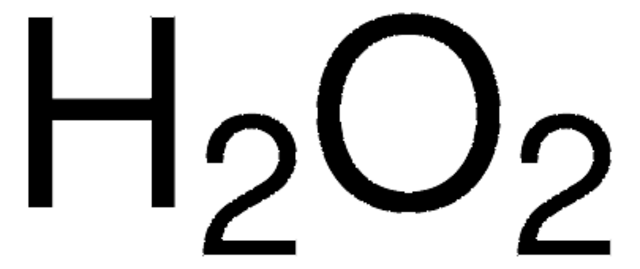P8412
o-Phenylenediamine dihydrochloride
tablet, 30 mg substrate per tablet
Synonym(s):
1,2-Phenylenediamine tablet, OPD, OPD Tablet
About This Item
Recommended Products
Quality Level
form
tablet
mp
258 °C (dec.) (lit.)
solubility
water: 1 tablet/10 mL, clear, colorless
storage temp.
2-8°C
SMILES string
Cl[H].Cl[H].Nc1ccccc1N
InChI
1S/C6H8N2.2ClH/c7-5-3-1-2-4-6(5)8;;/h1-4H,7-8H2;2*1H
InChI key
RIIWUGSYXOBDMC-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
- human immunoglobulin
- CTLA4Ig in serum samples
- albumin in culture medium
Reconstitution
signalword
Danger
Hazard Classifications
Acute Tox. 3 Oral - Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Aquatic Acute 1 - Aquatic Chronic 1 - Carc. 2 - Eye Irrit. 2 - Muta. 2 - Skin Sens. 1
Storage Class
6.1C - Combustible, acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Nitroblue Tetrazolium (NBT) is used with the alkaline phosphatase substrate 5-Bromo- 4-Chloro-3-Indolyl Phosphate (BCIP) in western blotting and immunohistological staining procedures. These substrate systems produce an insoluble NBT diformazan end product that is blue to purple in color and can be observed visually.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service