T7915
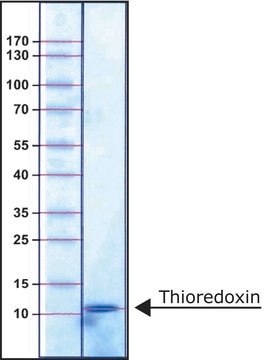
Thioredoxin Reductase from Escherichia coli
ammonium sulfate suspension, >25 units/mg protein (Bradford)
Synonym(s):
NADPH:oxidized thioredoxin oxidoreductase
About This Item
Recommended Products
biological source
Escherichia coli
Quality Level
form
ammonium sulfate suspension
specific activity
>25 units/mg protein (Bradford)
mol wt
70 kDa
technique(s)
activity assay: suitable
UniProt accession no.
storage temp.
2-8°C
Gene Information
Escherichia coli K12 ... trxB(949054)
General description
Thioredoxin reductases (TrxRs) belongs to family of selenium-containing pyridine nucleotide-disulphide oxidoreductases.
Application
- for determining the enzymatic activity of His6-Ahp1p.
- to assay C. elegans TRXR using the TXN-dependent activity assay.
- to investigate the biological reducing systems for organic hydroperoxide resistance R gene (OhrR).
- for enzymatic targeting of auranofin to test its antimicrobial properties.
- in thioredoxin reductase activity and peroxiredoxin assay.
- to study the synergy between broccoli sprout extract and selenium in the upregulation of thioredoxin reductase in human hepatocytes.
It has also been used to study the synergy between broccoli sprout extract and selenium in the upregulation of thioredoxin reductase in human hepatocytes.
Biochem/physiol Actions
Unit Definition
Physical form
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Instructions for working with enzymes supplied as ammonium sulfate suspensions
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service