40334
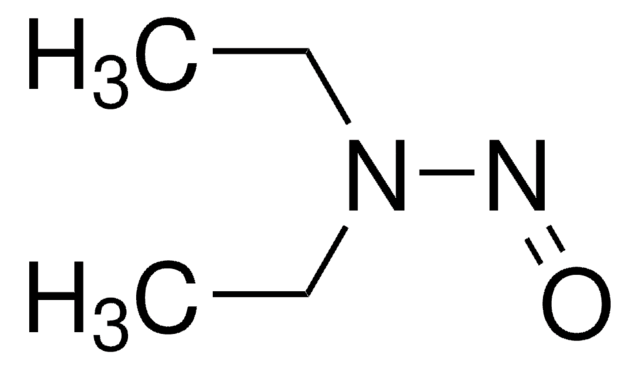
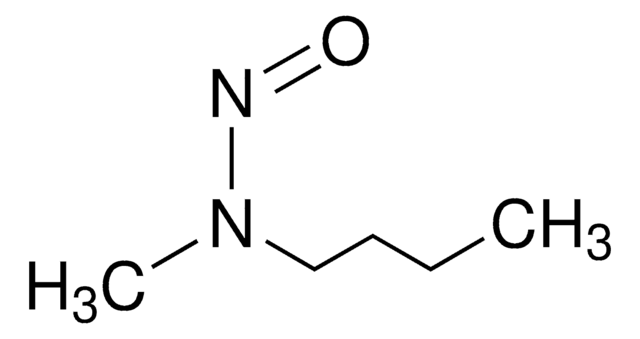
N-Nitrosodiethylamine (NDEA) solution
certified reference material, 5000 μg/mL in methanol
Synonym(s):
Diethylnitrosamine
About This Item
Recommended Products
grade
certified reference material
TraceCERT®
Quality Level
product line
TraceCERT®
CofA
current certificate can be downloaded
packaging
ampule of 1 mL
concentration
5000 μg/mL in methanol
technique(s)
HPLC: suitable
gas chromatography (GC): suitable
application(s)
cleaning products
cosmetics
environmental
food and beverages
personal care
format
single component solution
storage temp.
2-8°C
InChI
1S/C4H10N2O/c1-3-6(4-2)5-7/h3-4H2,1-2H3
InChI key
WBNQDOYYEUMPFS-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
N-Nitrosodiethylamine belongs to the group of toxic nitrosamines, known to show carcinogenic, mutagenic, and hepatotoxic properties. It is formed as a byproduct during food processing, pharmaceutical processing, and water treatment. As a result, they are generally found in water, fish, meat, drug substances, etc.
Application
- Simultaneous analysis of N-nitrosodiethanolamine, N-nitrosodiethylamine, triethanolamine, diethanolamine in cosmetic samples using liquid chromatography-electrospray ionization-tandem mass spectrometry (LC-ESI-MS/MS)
- Solid phase extraction of N-nitrosodimethylamine and N-nitrosomethylethylamine from eight sartans, ranitidine, and metformin and also the fortified products of these drugs, for quantification by gas chromatography-tandem mass spectrometry (GC-MS/MS)
- Multi-residue analysis of seven restricted nitrosamines in two body creams and two shower gel samples by vortex-assisted reversed-phase dispersive liquid-liquid microextraction (VA-RP-DLLME) and liquid chromatography-mass spectrometry (LC-MS)
- Quantitative analysis of four nitrosamines in four sartan drug substances— candesartan cilexetil, olmesartan medoxomi, irbesartan, and valsartan, by gas chromatography-tandem mass spectrometry (GC-MS/MS)
- Determination of eight volatile nitrosamines in 28 popular meat product samples using ultrasonic solvent extraction combined with solid phase microextraction (SPME) and GC-MS
Other Notes
Legal Information
signalword
Danger
Hazard Classifications
Acute Tox. 3 Dermal - Acute Tox. 3 Inhalation - Acute Tox. 3 Oral - Carc. 1B - Flam. Liq. 2 - STOT SE 1
Storage Class
3 - Flammable liquids
wgk_germany
WGK 3
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Learn about LC-MS/MS method development to quantify NDMA impurity in valsartan drug substance using Titan™ C18 column based UHPLC separation
Nitrosamines have been discovered as a serious contaminant group in active pharmaceutical ingredients (API) belonging to the sartan family. This article describes a GC-MS method for the determination of nitrosamines in Valsartan tablets according to US FDA guide lines that can be used for pharma QC.
Protocols
US EPA Method 8270 (Appendix IX): GC Analysis of Semivolatiles on Equity®-5 (30 m x 0.25 mm I.D., 0.50 μm)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service