BP849
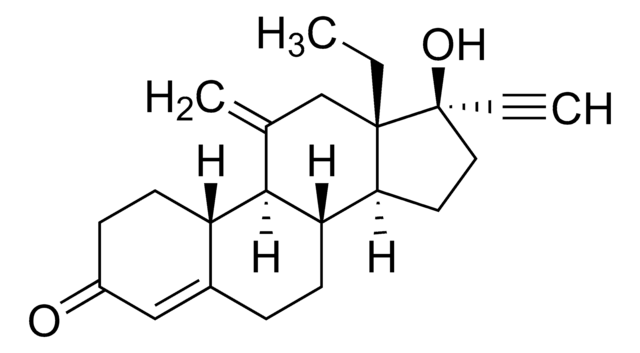
Tibolone impurity standard
British Pharmacopoeia (BP) Reference Standard
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
UNSPSC Code:
41116107
NACRES:
NA.24
Recommended Products
grade
pharmaceutical primary standard
API family
tibolone
form
solid
shelf life
limited shelf life, expiry date on the label
manufacturer/tradename
BP
application(s)
pharmaceutical
pharmaceutical small molecule
format
mixture
storage temp.
−20°C
General description
This product is provided as delivered and specified by the issuing Pharmacopoeia. All information provided in support of this product, including SDS and any product information leaflets have been developed and issued under the Authority of the issuing Pharmacopoeia. For further information and support please go to the website of the issuing Pharmacopoeia.
Application
Tibolone impurity standard, intended for use in laboratory tests only as specifically prescribed in the British Pharmacopoeia.
Also used in monographs such as:Tibolone Tablets
Also used in monographs such as:
Packaging
Unit quantity: 10 mg. Subject to change. The product is delivered as supplied by the issuing Pharmacopoeia. For the current unit quantity please visit British Pharmacopoeia
Other Notes
Sales restrictions may apply.
signalword
Warning
hcodes
Hazard Classifications
Eye Irrit. 2
wgk_germany
WGK 3
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Lot/Batch Number
Sorry, we don't have COAs for this product available online at this time.
If you need assistance, please contact Customer Support.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service