578878
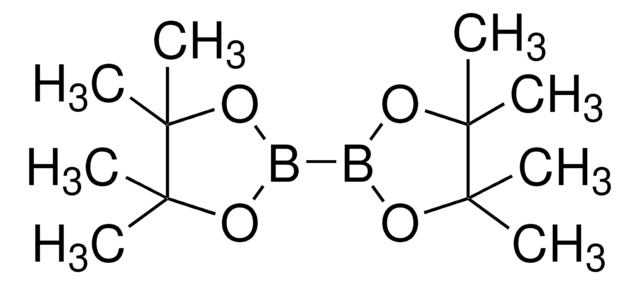
2,2′−Bithiophene-5-boronic acid pinacol ester
Synonym(s):
5-(4,4,5,5-Tetramethyl-1,3,2-dioxaborolan-2-yl)-2,2′-bithiophene
About This Item
Recommended Products
refractive index
n20/D 1.5900 (lit.)
Quality Level
mp
35.5-38.0 °C (average)
SMILES string
CC1(C)OB(OC1(C)C)c2ccc(s2)-c3cccs3
InChI
1S/C14H17BO2S2/c1-13(2)14(3,4)17-15(16-13)12-8-7-11(19-12)10-6-5-9-18-10/h5-9H,1-4H3
InChI key
HPOQARMSOPOZMW-UHFFFAOYSA-N
Related Categories
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Articles
Oligothiophenes are important organic electronic materials which can be produced using synthetic intermediates and Suzuki coupling.
This brochure contains a comprehensive selection of boronic acids, boronic acid esters, diboron esters, and transition-metal catalysts useful for the Suzuki–Miyaura coupling reaction
The synthesis of biaryl compounds via the Suzuki–Miyaura coupling reaction has become more commonplace now that many arylboronic acids are readily available.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service