D80002
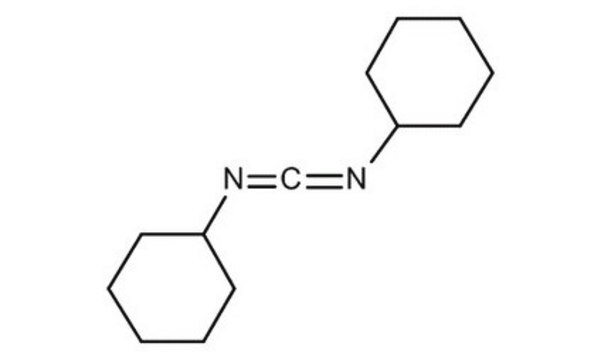
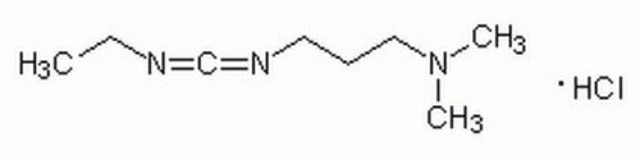
DCC
99%
Synonym(s):
N,N′-Dicyclohexylcarbodiimide
About This Item
Recommended Products
Quality Level
assay
99%
form
solid
reaction suitability
reaction type: Coupling Reactions
bp
122-124 °C/6 mmHg (lit.)
mp
34-35 °C (lit.)
SMILES string
C1CCC(CC1)N=C=NC2CCCCC2
InChI
1S/C13H22N2/c1-3-7-12(8-4-1)14-11-15-13-9-5-2-6-10-13/h12-13H,1-10H2
InChI key
QOSSAOTZNIDXMA-UHFFFAOYSA-N
Gene Information
human ... EPHX2(2053)
mouse ... Ephx2(13850)
Looking for similar products? Visit Product Comparison Guide
General description
Application
7-((tert-butyldimethylsilyl)oxy)hepta-2,4-diyn-1-ol with propiolic acid to form 7-((tert-butyldimethylsilyl)oxy)hepta-2,4-diyn-1-yl propynoate.
It can also used to synthesize:
- 1,3-Thiazetedine derivatives via [2+2] cycloaddition with 2-phenylethenyl- and 2-(4-nitrophenyl)ethenyl isothiocyanates.
- 1,3,5-Oxadiazine-4-thiones via [4+2] cycloaddition with benzoyl isothiocyanates.
- Sterically hindered 1,3,4-oxadiazole derivatives by reacting with (N-isocyanimino)triphenylphosphorane in the presence of aromatic (or heteroaromatic) carboxylic acids.
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 3 Dermal - Acute Tox. 4 Oral - Eye Dam. 1 - Skin Sens. 1
wgk_germany
WGK 3
flash_point_f
closed cup
flash_point_c
closed cup
ppe
Eyeshields, Faceshields, Gloves, type P2 (EN 143) respirator cartridges
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Carbodiimide-mediated peptide coupling remains to the most frequently used technique.
Amide bonds are ubiquitous in both nature and industrial applications. They are vital to the structure and function of biological macromolecules and polymers. The importance of this functionality has resulted in numerous approaches to its formation, ranging from stoichiometric activation of carboxylic acids to more recent advances in catalytic amide bond formation.
Professor Aran (Claremont University, USA) thoroughly discusses the engineering of graphene based materials through careful functionalization of graphene oxide, a solution processable form of graphene.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service