233153-M
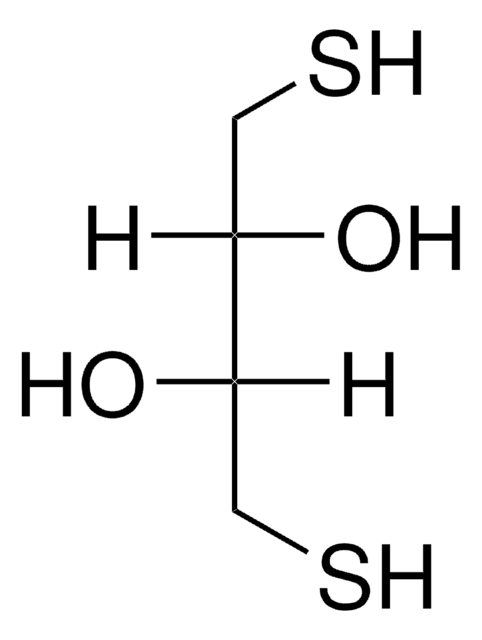
Cleland′s Reagent
ULTROL® Grade, ≥99% (titration), protective agent for SH groups
Synonym(s):
Cleland’s Reagent, ULTROL® Grade, Dithiothreitol, DTT
About This Item
Recommended Products
Quality Level
assay
≥99% (titration)
form
solid
reaction suitability
reagent type: reductant
manufacturer/tradename
Calbiochem®
storage condition
OK to freeze
color
white
solubility
water: 200 mM
ethanol: soluble
cation traces
heavy metals: <1 ppm
storage temp.
2-8°C
InChI
1S/C4H10O2S2/c5-3(1-7)4(6)2-8/h3-8H,1-2H2/t3-,4-/m0/s1
InChI key
VHJLVAABSRFDPM-IMJSIDKUSA-N
Related Categories
General description
Application
- Mechanochemical disulfide reduction reveals imprints of noncovalent sulfuroxygen chalcogen bonds in protein-inspired mimics in aqueous solution.: This study highlights the use of Cleland′s reagent in exploring noncovalent interactions in protein mimics under mechanochemical conditions, shedding light on disulfide bond dynamics relevant to protein engineering and drug design (Dopieralski et al., 2020).
- A Simple In Vitro Gut Model for Studying the Interaction between Escherichia coli and the Intestinal Commensal Microbiota in Cecal Mucus.: Utilizes Cleland′s Reagent in microbiological assays to understand the complex interactions within the gut microbiome, providing insights important for developing therapeutic strategies against intestinal diseases (Mokszycki et al., 2018).
- Synthesis of the marine bromotyrosine psammaplin F and crystal structure of a psammaplin A analogue.: Discusses the role of Cleland′s reagent in the synthesis and structural analysis of marine natural products, which are potential leads for novel pharmaceuticals (Yang et al., 2010).
- Effects of dithiothreitol on protein activity unrelated to thiol-disulfide exchange: for consideration in the analysis of protein function with Cleland′s reagent.: This research investigates the broader biochemical impacts of Cleland′s Reagent (dithiothreitol) beyond its primary use, offering a cautionary perspective for its application in protein studies (Alliegro, 2000).
- A modified reagent for the confirmation of blood.: Describes a forensic application where Cleland′s Reagent is modified for enhanced sensitivity and specificity in blood detection, crucial for forensic science and criminal investigations (Hatch, 1993).
Warning
Reconstitution
Other Notes
Lauriault, V.V.M., and O′Brien, P.J. 1991. Mol. Pharmacol.40, 125.
Klonne, D.R., and Johnson, D.R. 1988. Toxicol. Lett.42, 199.
Zhang, R., and Snyder, G.H. 1988. Biochemistry27, 3785.
Shaked, Z., et al. 1980. Biochemistry19, 4156.
Zahler, W.L., and Cleland, W.W. 1968. J. Biol. Chem.243, 716.
Cleland, W.W. 1964. Biochemistry3, 480.
Legal Information
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Eye Dam. 1 - Skin Irrit. 2
wgk_germany
WGK 2
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service