T2879
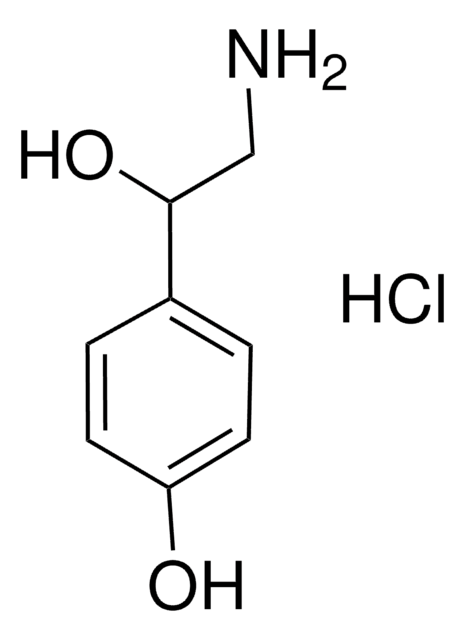
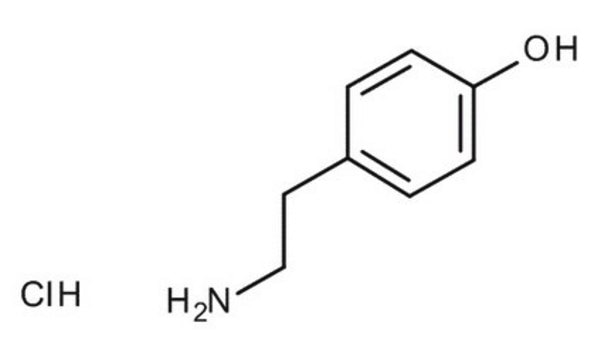
Tyramine hydrochloride
≥98%
Synonym(s):
4-(2-Aminoethyl)phenol hydrochloride, 4-Hydroxyphenethylamine hydrochloride, Tyrosamine hydrochloride
About This Item
Recommended Products
Quality Level
assay
≥98%
mp
271-274 °C (lit.)
solubility
H2O: soluble 50 mg/ml, clear, colorless to faintly yellow
SMILES string
Cl[H].NCCc1ccc(O)cc1
InChI
1S/C8H11NO.ClH/c9-6-5-7-1-3-8(10)4-2-7;/h1-4,10H,5-6,9H2;1H
InChI key
RNISDHSYKZAWOK-UHFFFAOYSA-N
Gene Information
human ... ADORA2A(135) , ADORA2B(136) , ADORA3(140)
Looking for similar products? Visit Product Comparison Guide
Application
- coinfused with adenosine in control subjects and patients in order to reduce leg blood flow by 50% without affecting arterial blood pressure
- labelled with fluorescence dyes (Atto 488 and Atto 655) to serve as a substrate for peroxidase in immunofluorescence analysis
- used in dimethylformamide, labelled with 5-(and-6)carboxyfluorescein, succinimidyl ester/biotin to serve as a substrate for peroxidase in tyramide signal amplification
Biochem/physiol Actions
related product
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service